Supplement Labeling Statements
With numerous regulations in place, ensuring that products meet regulatory guidelines for supplement labeling can be cumbersome. Clear and accurate product labeling is not only an FDA requirement but is an essential component to product marketing and is crucial for providing consumers with necessary information about the supplements they buy. Among the FDA supplement labeling regulations, there are five fundamental statements that every dietary supplement label must prominently display.
- Statement of Identity
The statement of identity serves as the primary introduction to a product. It is, in essence, the name of the dietary supplement. Whether presenting a “Vitamin C tablet” or “Omega-3 Fish Oil,” this identity needs to be distinctly evident on the product. Consistency is crucial in this statement. Ensure that this name seamlessly aligns with the terminology used in other marketing materials.
Tip for Manufacturers: Ensure that this name seamlessly aligns with the terminology used in other marketing materials. Testing for Identify is a key component of the FDA 21 CFR part 111 regulation of supplements which aims to ensure the quality of the dietary supplements and to ensure that the dietary supplements are packaged and labeled as specified in the master manufacturing record.
- Net Quantity of Contents Statement
The new quantity of contents statement offers consumers clarity about the amount of the dietary supplement in their purchase. Whether the product measures in tablets, capsules, milliliters, or other units, the listed quantity should be precise. This transparency not only meets consumer expectations but also ensures regulatory compliance.
Tip for Manufacturers: Precision is essential. Ensure that your manufacturing processes align with the stated quantity to meet both consumer expectations and regulatory compliance.
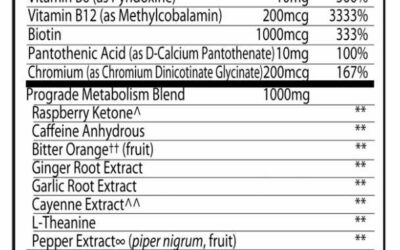
- Nutrition Labeling also known as Supplement Fact Panel
More commonly known as the “Supplement Facts” panel, this section details the dietary ingredients contained within the supplement. Information will be displayed in a panel format and will list aspects such as serving size, active ingredients, and their respective quantities, along with other pertinent nutritional information.
Tip for Manufacturers: Regularly review and verify the accuracy of the information, especially when there are changes in the formulation or ingredient sourcing. This should be done through ongoing testing of your raw materials (ingredients) to make sure they are meeting your potency requirements and supporting any claims you are making on your package.
- Ingredient List
Beyond the primary dietary ingredients highlighted in the supplement facts panel, the ingredient list provides a breakdown of all components in the supplement. This includes inactive ingredients, such as fillers, binders, colors, and any other compounds used.
Tip for Manufacturers: It is standard practice to list these ingredients in descending order based on weight, ensuring transparency about the supplement’s composition.
- Name and Place of Business
Every dietary supplement label must include the name and place of business of the manufacturer, packer, or distributor. This ensures accountability and offers consumers a point of contact should they have questions or concerns.
Tip for Manufacturers: Make sure the provided details are current, especially if there are any changes in business locations or ownership. Consider also including contact information, such as a phone number or website, to enhance consumer trust.
Placement of Label Statements
Supplement labels have two areas to display information, the front of the label panel also known as the principal display panel, and the information panel which is usually immediately to the right of the principal display panel. The information panel must include the supplement facts panel, the ingredient list, and the name and place of business. All other required label statements can appear in either location.
Labeling in the dietary supplement domain is not just about meeting regulatory requirements. It’s a manifestation of a manufacturer’s dedication to transparency, quality, and consumer trust. By meticulously integrating these five statements into your product labels, you not only align with FDA guidelines, but you also elevate your brand’s reputation in a market that values transparency.